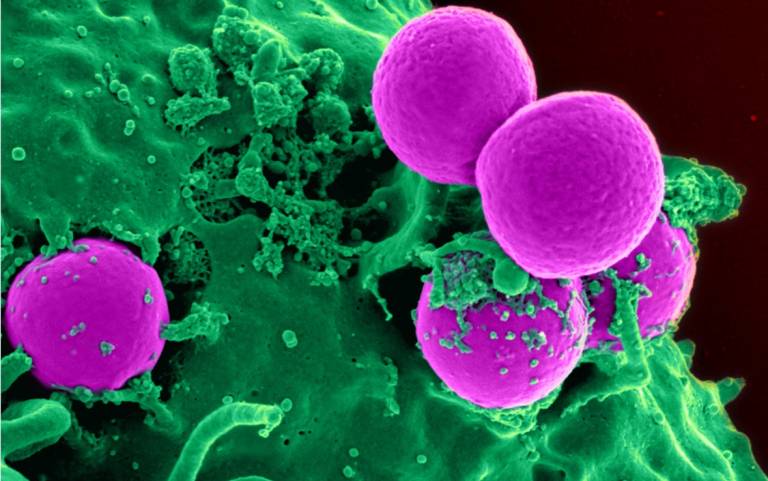
Light-activated antibacterial coatings are nothing new, but photobactericidal activity generally requires UV light or intense white light in the region of 3,000 lux. The UCL team claims its research, published in Nature Communications, describes the first coating to demonstrate antibacterial properties under ambient light conditions of around 300 lux.
Layered Manuka honey helps battle bacteria
New bandage clots blood but doesn’t stick to wound
“Other coatings have effectively killed bacteria but only after exposure to UV light, which is dangerous to humans, or very intense light sources, which aren’t very practical,” said senior author Professor Ivan Parkin, Dean of UCL Mathematical & Physical Sciences. “We are surprised to see just how effective our coating is in killing both S. aureus (MRSA) and E. coli in ambient light, making it promising for use in a variety of healthcare environments.”
The bactericidal coating consists of a polymer embedded with crystal violet - a dye with antibacterial and antifungal properties – and thiolated gold nanoclusters. Under illumination of 312 lux, the team demonstrated that a polymer with crystal violet alone did not kill bacteria. However, the addition of the gold nanoclusters saw hydrogen peroxide produced under this low light, which broke down the cell walls of the bacteria.
“Dyes such as crystal violet are promising candidates for killing bacteria and keeping surfaces sterile as they are widely used to disinfect wounds,” said first author, Dr Gi Byoung Hwang, from UCL Chemistry.
“When exposed to bright light, they create reactive oxygen species, which in turn kill bacteria by damaging their protective membranes and DNA. This is amplified when they are paired with metals such as silver, gold and zinc oxide.”
The coating led to a 3.3 log reduction in the growth of MRSA after six hours and a 2.8 log reduction in the growth of E. coli after 24 hours. According to senior author Professor Asterios Gavriilidis, from UCL Chemical Engineering, only a tiny amount of gold is required in order to facilitate the reaction.
“The gold clusters in our coating are key to generating the hydrogen peroxide, through the action of light and humidity,” he said.
“Given the clusters contain only 25 atoms of gold, very little of this precious metal is required compared to similar coatings, making our coating attractive for wider use.”




Swiss geoengineering start-up targets methane removal
No mention whatsoever about the effect of increased methane levels/iron chloride in the ocean on the pH and chemical properties of the ocean - are we...