
Charge storage devices, such as super capacitors and batteries, are often made from metal oxides with a tunnel-like structure. It has been known for some time that incorporating large metal ions, such as potassium, into the structure of these tunnels can improve the material’s performance, but material scientists and chemists have not understood why this is the case. A team led by Prof Saiful Islam of Bath, working with Prof Reza Shahbazian-Yassar of the University of Illinois at Chicago, have used a combination of structural experiments and computer simulation to investigate the phenomenon.
In a paper in Nature Communications, the team explains that previous theories have stated that positive ions (or cations) block lithium ions from travelling down the tunnels, forcing them into the structure of the device; others believe that the cations stop the tunnels from collapsing.
Using manganese dioxide as a study material, the team looked at how doping the structure with potassium affected lithium transport. They found that rather than blocking the ions, cation doping actually helps charge carriers move faster through the structure, improving conductivity and diffusivity of the material. Moreover, the tunnels remain intact whether cations are present or not.
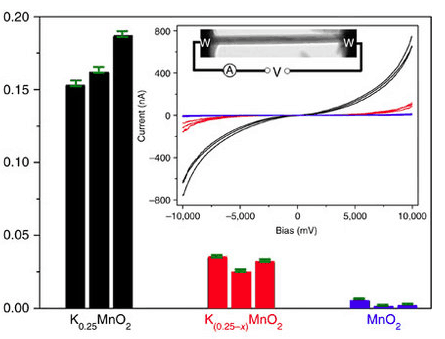
"Understanding these processes is important for future design and development of battery materials, and could lead to faster charging batteries that will benefit consumers and industry," said Prof Islam. "Developing new materials holds the key to lighter, cheaper and safer batteries, including for electric vehicles that will cut carbon emissions."




Nanogenerator consumes CO2 to generate electricity
Whoopee, they've solved how to keep a light on but not a lot else.