Ben Wiley, a professor of chemistry at Duke University, North Carolina and his team recently tested three new materials that might be used as a porous, flow-through electrode to improve the efficiency of electrolysis. Their goal was to increase the surface area of the electrode for reactions, while avoiding trapping the gas bubbles that are produced.
HyNet hydrogen project gets £13m funding boost
"The maximum rate at which hydrogen is produced is limited by the bubbles blocking the electrode - literally blocking the water from getting to the surface and splitting," Wiley said in a statement.
In a paper published in Advanced Energy Materials, they compared three different configurations of a porous electrode through which the alkaline water can flow as the reaction occurs.
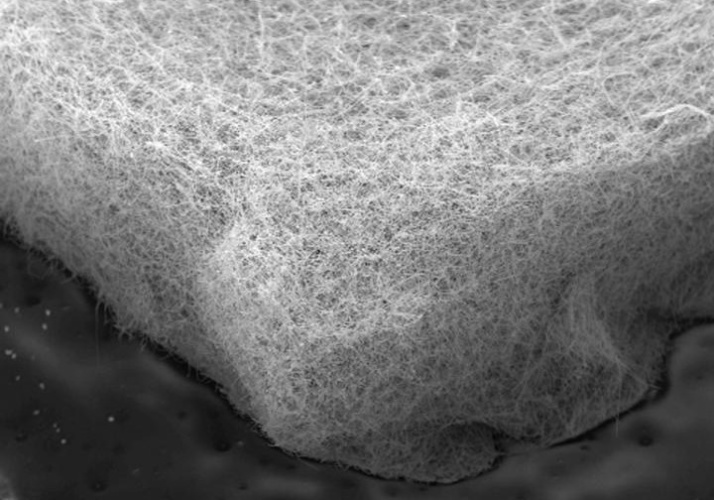
They fabricated three kinds of flow-through electrodes, each a 4mm square of sponge-like material, just 1mm thick. One was made of a nickel foam, one was a 'felt' made of nickel microfibres, and the third was a felt made of nickel-copper nanowires.
Pulsing current through the electrodes for five minutes on, five minutes off, they found that the felt made of nickel-copper nanowires initially produced hydrogen more efficiently because it had a greater surface area than the other two materials. But within 30 seconds, its efficiency plunged because the material got clogged with bubbles.
The nickel foam electrode was best at letting the bubbles escape, but it had a significantly lower surface area than the other two electrodes, making it less productive.
A felt of nickel microfibre produced more hydrogen than the nanowire felt, despite having 25 per cent less surface area for the reaction.
Over the course of a 100-hour test, the microfibre felt produced hydrogen at a current density of 25,000 milliamps per square centimetre. At that rate, it would be 50 times more productive than the conventional alkaline electrolysers currently in use for water electrolysis, the researchers calculated.
The cheapest way to make industrial quantities of hydrogen involves breaking methane apart with steam, which is an energy-intensive approach that creates 9 to 12 tons of C02 for every ton of hydrogen it yields, not including the energy needed to create 1000oC steam.
Wiley said commercial producers of water electrolysers may be able to make improvements in the structure of their electrodes based on what his team has learned. If they could greatly increase the hydrogen production rate, the cost of hydrogen produced from splitting water could go down, perhaps even enough to make it an affordable storage solution for renewable energy.
He is also working with a group of students in Duke's Bass Connections program who are exploring whether flow-through electrolysis might be scaled up to make hydrogen from India's abundant solar power.




Nanogenerator consumes CO2 to generate electricity
Whoopee, they've solved how to keep a light on but not a lot else.