The project, called CL4W (Cleaning Land for Wealth, pronounced ‘claw’) could help develop cheaper and lower-energy ways of remediating contaminated land as well as producing valuable commercial products, said project leader Kerry Kirwan.
The team, which includes researchers from the Universities of Edinburgh, Newcastle, Cranfield and Birmingham, came together as part of an EPSRC ‘sandpit’ to pool resources on biorefining and synthetic biology.
‘We’ve known for many years that plants can suck up metals from the soil; it’s why bananas are rich in potassium and cabbage has iron,’ Kirwan told The Engineer.
‘And bioremediation is well established — it was used to clean up some land around Chernobyl using hemp and flax, for example. But we had a good look at the literature, and it seems that nobody has tried to recover the metals from the plants. They generally just burn them, or if the metal is really toxic or radioactive, lock them away safely.’
Warwick already has expertise in techniques which break down the tough lignocellulose structure of plants, Kirwan explained. ‘We’ve demonstrated that we can use microbes to do that and produce compounds such as polymer feedstocks at relatively low energy,’ he said. Using this technique would produce a culture of microbes that incorporate the metal ions from plants grown on contaminated land.
The next stage would be to feed this culture to another group of bacteria which have the ability to reduce metal ions to their solid metallic form. ‘A lot of bacteria have this ability — it’s their defence mechanism against toxic metal ions,’ explained Louise Horsfall of Edinburgh University, a researcher in synthetic biology.
‘Some of them excrete the metal inside their cells as nanocrystals — you can find perfect cubes of gold or triangular prisms of silver in some of them. Others reduce them to the metal outside their cell wall, and produce cruder blobs of metal which can form a kind of shell.’
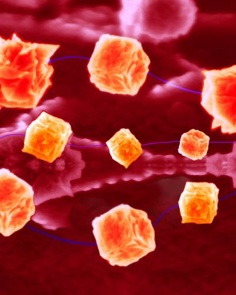
Using synthetic biology, an array of genetic techniques to produce biological effects not found in nature, Horsfall believes that it will be possible to tailor the shapes of the nanoparticles produced by metal-secreting bacteria to produce specific properties needed by different branches of industry.
‘You can’t make a crystal take up a different form than it would take in nature, but we could choose the size of the faces of the crystal, for example,’ she said. ‘But we wouldn’t be introducing any modified bacteria into the environment; it would only be natural plants we’d grow on the contaminated land, and any modified bacteria would be used in safe, controllable enclosed reactors.’
Kirwan elaborated: ‘There are some bacteria which produce a spherical shell under some conditions, but if you change the conditions, they take up a different shape. We might be able to make spheres, or long cylinders, by choosing which bacteria and selecting the conditions we grow them in.’
The team decided to concentrate their efforts on plants which incorporate platinum group metals (PGMs) and arsenic into their tissues. There are a great many of these which grow in different environments all around the world, Kirwan said, such as the common flower group Alyssum which has hundreds of sub-species; this would mean that a new species would not have to be introduced to an area to clean a site.
There is a growing problem of platinum-group pollution in riverbanks near electronics production facilities, he said, and platinum has also been seen to accumumate in the environment owing to dust from catalytic converters. Arsenic, meanwhile, is also a byproduct of industrial processes, and also occurs naturally.

Moreover, PGMs and arsenic are both valuable commodities; the former as industrial catalysts, while platinum and arsenic nanoparticles are used to treat aggressive cancers. ‘These are high-price products,’ Kirwan said. ‘This would give manufacturers of catalytic convertors, developers of cancer treatments and other applicable technologies exactly the right shape, size and functionality they need without subsequent refinement.’
The advantage of this technique would be that it doesn’t require energy-intensive, costly procedures to clean land. ‘We are saying that we can use natural processes to help things along,’ Kirwan said. ‘It could also be used to recover metals from landfills or from slag heaps, or even to harvest the last traces of metals from open-cast mines.’
The project, which will last for four years, has received £3m of EPSRC funding; by the end, Kirwan hopes to demonstrate the whole process, from identifying suitable sites and remediation plant species, to producing tailored metal nanoparticles. ‘We are also expecting to recover other high value materials such as fine chemicals, pharmaceuticals, anti-oxidants etc. from the crops during the same biorefining process.’




Glasgow trial explores AR cues for autonomous road safety
They've ploughed into a few vulnerable road users in the past. Making that less likely will make it spectacularly easy to stop the traffic for...