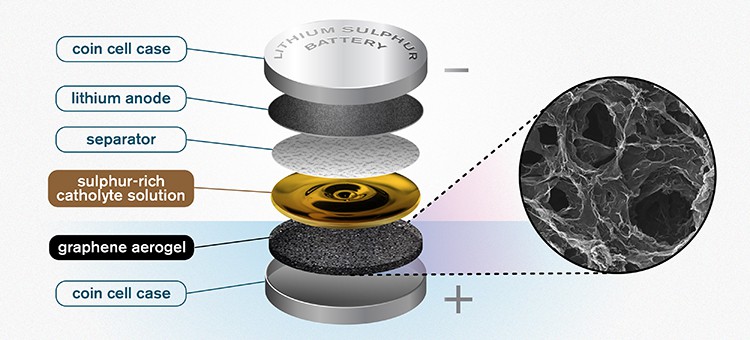
Developed at Chalmers University of Technology in Gothenburg, the prototype consists of slices of porous graphene oxide aerogel in a cell battery that soak up a sulphur-rich ‘catholyte’ solution. This catholyte acts as cathode and electrolyte, interacting with a lithium anode via a separator layer within the cell. The concept helps save weight and can deliver faster charging and better power output.
“You take the aerogel, which is a long thin tube, and then you slice it – almost like a salami,” said Carmen Cavallo, lead researcher on the study, which appears in the Journal of Power Sources. “You take that slice, and compress it, to fit into the battery.”
The sulphur-rich catholyte solution is then added, with the highly porous aerogel acting as the support, soaking up the liquid like a sponge.

“The porous structure of the graphene aerogel is key,” Cavallo continued. “It soaks up a high amount of the catholyte, giving you high enough sulphur loading to make the catholyte concept worthwhile. This kind of semi-liquid catholyte is really essential here. It allows the sulphur to cycle back and forth without any losses. It is not lost through dissolution – because it is already dissolved into the catholyte solution.”
With Li-ion technology approaching its theoretical performance limit, Li-S has emerged as a leading candidate to replace it. Current Li-ion batteries operate at about 300W h/kg (Watt-hours per kg), with a notional maximum of around 350. By comparison, Li-S batteries have a theoretical ceiling of between 1,000-1,500W h/kg.
“Furthermore, sulphur is cheap, highly abundant, and much more environmentally friendly,” said Aleksandar Matic, a Professor at Chalmers Department of Physics and head of the research group behind the paper. “Lithium sulphur batteries also have the advantage of not needing to contain any environmentally harmful fluorine, as is commonly found in lithium-ion batteries.”
One major problem hindering adoption has been the relative instability of Li-S batteries and their subsequent low life-cycle, making them unsuitable for electronic devices such as smartphones. The new design avoids two main problems with the degradation of Li-S batteries: firstly, that the sulphur dissolves into the electrolyte and is lost, and second, a ‘shuttling effect’ whereby sulphur molecules migrate from the cathode to the anode. In this design, these effects can be drastically reduced. Getting the technology to market will not be simple, however.
"Since these batteries are produced in an alternative way from most normal batteries, new manufacturing processes will need to be developed to make them commercially viable," said Matic.




Swiss geoengineering start-up targets methane removal
No mention whatsoever about the effect of increased methane levels/iron chloride in the ocean on the pH and chemical properties of the ocean - are we...