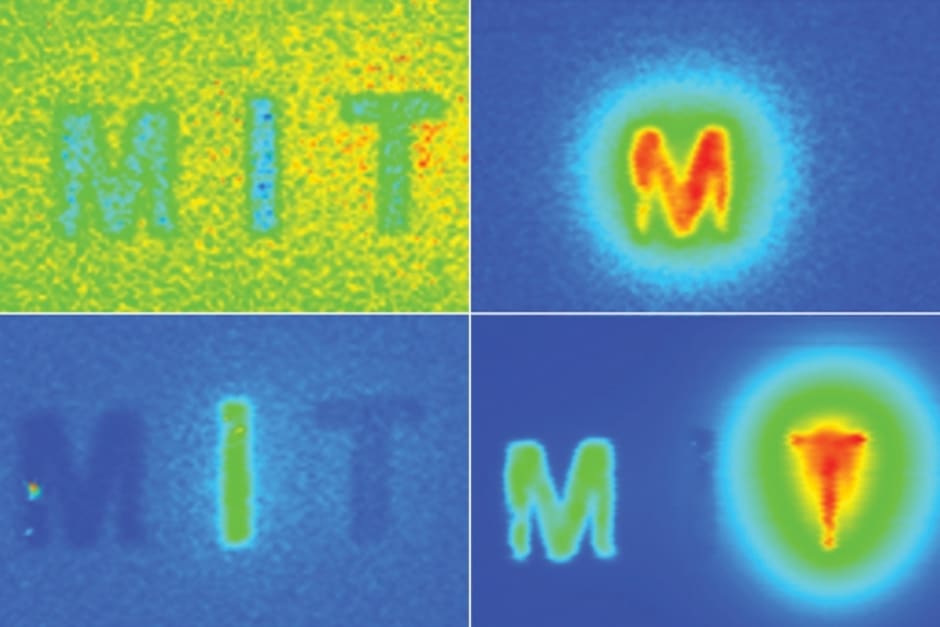
The researchers used DOLPHIN (Detection of Optically Luminescent Probes using Hyperspectral and diffuse Imaging in Near-infrared) to track a 0.1mm fluorescent probe through the digestive tract of a living mouse. They also showed that they can detect a signal to a tissue depth of 8cm, which is said to be far deeper than any existing biomedical optical imaging technique.
The researchers hope to adapt their imaging technology for early diagnosis of ovarian and other cancers that are currently difficult to detect until late stages.
"We want to be able to find cancer much earlier," said Angela Belcher, the James Mason Crafts Professor of Biological Engineering and Materials Science at MIT and a member of the Koch Institute for Integrative Cancer Research, and the newly-appointed head of MIT's Department of Biological Engineering. "Our goal is to find tiny tumours and do so in a non-invasive way."
Existing methods for imaging tumours have a trade-off between resolution and depth of imaging, and none of the optical imaging techniques can image deeper than about 3cm into tissue. Commonly used scans such as X-ray computed tomography (CT) and magnetic resonance imaging (MRI) can image through the whole body but they can't reliably identify tumours until they reach about 1cm in size.
Near-infrared light, which has wavelengths from 900 to 1700nm, is suited to tissue imaging because light with longer wavelengths doesn't scatter as much as when it strikes objects, which allows the light to penetrate deeper into the tissue. To take advantage of this, the researchers used hyperspectral imaging, which enables simultaneous imaging in multiple wavelengths of light.
The researchers tested their system with a variety of near-infrared fluorescent light-emitting probes, mainly sodium yttrium fluoride nanoparticles that have rare earth elements such as erbium, holmium, or praseodymium added through doping. Depending on the choice of the doping element, each of these particles emits near-infrared fluorescent light of different wavelengths.
The researchers developed algorithms that helped them analyse the data from the hyperspectral scan to identify the sources of fluorescent light of different wavelengths, which allows them to determine the location of a particular probe. By further analysing light from narrower wavelength bands within the entire near-IR spectrum, the researchers can also determine the depth at which a probe is located.
To demonstrate the potential usefulness of this system, the researchers tracked a 0.1mm-sized cluster of fluorescent nanoparticles that was swallowed and then travelled through the digestive tract of a living mouse. These probes could be modified so that they target and fluorescently label specific cancer cells.
"In terms of practical applications, this technique would allow us to non-invasively track a 0.1mm-sized fluorescently-labelled tumour, which is a cluster of about a few hundred cells. To our knowledge, no one has been able to do this previously using optical imaging techniques," said Neelkanth Bardhan, a Mazumdar-Shaw International Oncology Fellow.
Belcher is the senior author of the study, which appears in Scientific Reports. Bardhan and Xiangnan Dang, a former MIT postdoc, are the lead authors of the study. Other authors include research scientists Jifa Qi and Ngozi Eze, former postdoc Li Gu, postdoc Ching-Wei Lin, graduate student Swati Kataria, and Paula Hammond, the David H. Koch Professor of Engineering, head of MIT's Department of Chemical Engineering, and a member of the Koch Institute.




Glasgow trial explores AR cues for autonomous road safety
They've ploughed into a few vulnerable road users in the past. Making that less likely will make it spectacularly easy to stop the traffic for...