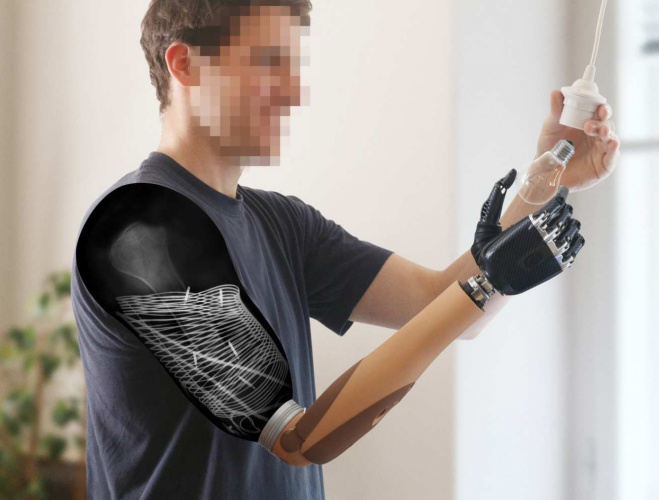
For amputees who use prosthetics, control of their robotic limb is an important factor for their quality of life. The closer medical engineers can get to the function of a natural limb, the more comfortable the amputee tends to be, and comfort tends to correlate to confidence in using the prosthetic in everyday life.
Bioengineers at Imperial College London, collaborating with colleagues in the Medical University of Vienna, carried out a small-scale test on a group of amputees who had lost an arm above the elbow, and reported encouraging results in Science Robotics.
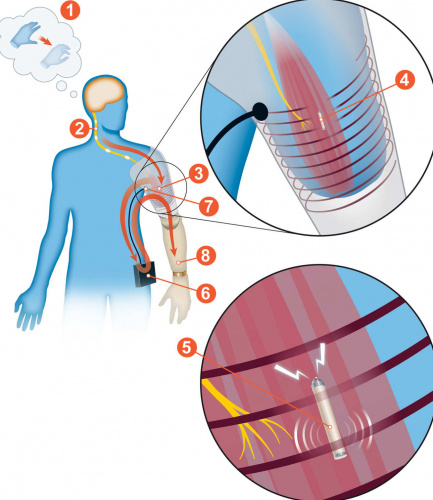
The research, which was led by the Vienna team, involved relocating nerves in the stumps of three patients, implanting wirelessly-chargeable electrodes and fitting new prostheses. In the paper, the team reports the patients found that, in the two years after the surgery, they could move their arms more easily and with better accuracy and precision.
Nadine Stech on designing the world’s most advanced prosthetic leg
Future prosthetic: towards the bionic human
The project aimed to improve on current prosthetic control, which works using sensors in the socket of the prosthetic to detect subtle movements in the muscles of the residual upper arm and convert these signals into the movements of the robotic arm and hand.
Similar systems work in prostheses for lower limb amputees to work knee, ankle and foot joints. However, slippage of the socket, moisture from sweat, and swelling of tissues can all disrupt detection and pickup of signals between muscles and sensors.
Implanting electrodes into the stump to connect directly with the prosthetic has been considered before, but even as technology has improved, many surgeons have hesitated to suggest this as amputees have often had traumatic experiences and further surgery has been believed to be an unattractive option.
The Imperial and Vienna teams have found that, despite the need for extra surgery, the ability to control a prosthetic better has in fact reduced rehabilitation times.
Their technique involves connecting electrodes directly to nerves and muscles in the arm stump, allowing the prosthetic to “feel” deeper muscles to get a better idea of the precise movements wanted by the user.
As well as improving control, this made the fitting of the prosthetic socket – conventionally a time-consuming and uncomfortable process which has to be repeated
regularly – much easier, as there is no need to place surface electrodes between sockets and the skin of the residual limb. There was also less rubbing, and sweating did not reduce the level of control of quality and precision of movements.
“Although this was demonstrated in only three people, the results are promising. From here, we hope to test the technique on more amputees to refine our method and make sure it’s fit to be used in more people with amputations,” commented Prof Dario Farina of Imperial College, a co-author on the paper. “Current prosthetics have come a long way since their inception. However, there’s still a lot of room for improvement.”
“We wanted to address the issues that cause frustration and even embarrassment to users, and ultimately improve amputees’ quality of life.”
The three subjects of the trial were men aged 16, 31 and 47. The surgery needed for the project involved relocating nerves within the stump of the residual arms and implantation of wirelessly-chargeable electrodes. This is the first long-term study combining implantable systems with nerve transfer surgery.
“We know that in healthy individuals, feedback from the prosthetic is very important for amputees to perceive the limb as a part of their own body. We hope our work here and in the future will go some way to make this happen,” said Prof Farina.
All three subjects reported preferring the new system to conventional prosthetics and wanted to keep their prostheses after the experimental period ended, as they worked well in the clinic and at home.




April 1886: the Brunkebergs tunnel
First ever example of a ground source heat pump?