When five-year-old Alex Barnes was diagnosed with an aggressive brain tumour the conventional options were bleak: surgery, followed by radiotherapy, with a strong chance of serious brain damage.
Fortunately, there was another choice. Thanks to the generosity of friends and relatives, the parents of the Leicestershire child were able to afford to take him to the US to receive proton therapy: a highly targeted, particle physics-based treatment that has a number of compelling advantages over traditional non-invasive approaches. In September 2010, two years after his treatment, Alex was given the all-clear and doctors believe there is now little chance of the tumour returning.
Proton therapy is one of a suite of socalled particle-therapy treatments that use equipment more readily associated with large physics experiments to hurl highly energised atomic particles or ions at cancer cells with pinpoint accuracy.
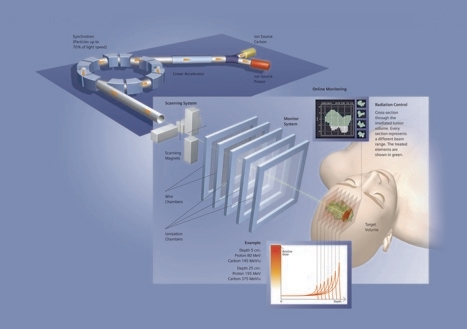
Claimed to be more effective than radiotherapy and with fewer unpleasant side effects, particle therapy is increasingly seen as one of the most promising weapons in the fight against cancer.
Although the treatment has been available for a number of years in various research clinics, the technology now seems to be moving into the medical mainstream. Indeed, just last month, the Department of Health announced that three UK hospitals — the Christie in Manchester, University College London Hospital and Birmingham University Hospital — have been shortlisted as possible sites for proton-beam facilities. A smaller low-energy facility has been operating at Clatterbridge hospital on the Wirral since the mid-1980s but this is only suitable for treating eye tumours. Meanwhile, around the world a number of dedicated, full-scale systems are in various stages of development.
One of the main firms spearheading this growth is Siemens Healthcare, which is currently building two dedicated facilities in Germany and one in Shanghai.

Based on a prototype system in Heidelberg that has been treating patients since last November, the first of these facilities, built in Marburg, Germany, will be handed over to the customer, German private-hospital group RKA, in the middle of next year. It is expected to begin treating patients around six months later.
Roughly the size of a football pitch, Siemens’ so-called Iontris system uses a linear accelerator and a synchrotron to accelerate protons to 250MeV and ionised carbon atoms to 430MeV before sending them down beam-lines to carefully positioned robotic treatment tables.
These pulsed beams of energetic particles, moving through the synchrotron at roughly 50 per cent of the speed of light, can be very precisely optimised accordingto the treatment required. For instance, while the energy level determines penetration depth, the intensity determines the irradiance and the focus determines the decrease in dosage for the surrounding healthy tissue.
According to Lucy Attwood, Siemens’ UK particle-therapy manager, the approach has a number of advantages over traditional radiotherapy.
Firstly, because the treatment is highly targeted, it is possible to define precisely where in the body the energy is deposited. ‘With normal radiotherapy they bombard the body with gamma rays that ionise everything in their path, destroying everything as they go — both healthy cells and cancer cells. This method literally just destroys a specific area,’ explained Attwood. Because fewer healthy cells are destroyed, the treatment is less painful and has fewer immediate side effects than radiotherapy.
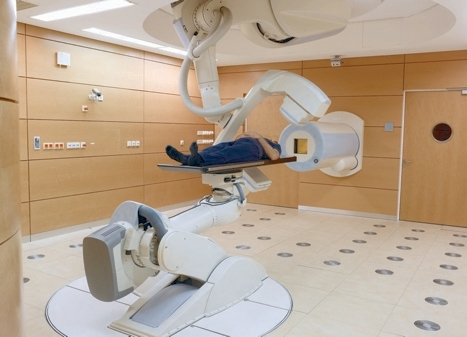
More importantly, it is also thought to reduce the likelihood of secondary cancer taking hold. ‘The main long-term side effect of radiotherapy is that the chance of getting secondary cancer is increased. That is dramatically reduced with particle therapy, as it does not damage the surrounding area.’
Attwood added that the treatment is particularly well suited for treating patients with tumours close to vital organs, which could be badly damaged by high doses of radiotherapy. ‘Say for example someone has a tumour in their eye,’ she said. ‘With normal radiotherapy, treating the tumour may also blind the patient. Localised particle therapy damages only the tumour.’ But despite the technology’s huge promise, there are limitations — not leastthe cost. Indeed, according to Attwood, a Siemens’ Iontris system costs around €250m and must treat around 2,000 patients a year to be economically viable.
The technology is also somewhat held back by its size. ‘A particle accelerator has to be the size of a football pitch to work, so it’s not something you can just bolt onto any hospital,’ said Attwood, adding that a longterm aim is to reduce the system’s footprint. Future versions are also likely to make the technology applicable to tumours near permanently mobile organs such as the heart.
Cyclotron-based systems
A US company is pursuing the same aim with an alternative technology
Siemens’ biggest competitor in the particle-therapy field is US firm IBA, which claims to have sold 18 proton therapy systems worldwide.
While Siemens’ Iontris system is based on a synchrotron, which synchronises the magnetic and electric fields, IBA’s technology uses a different particle accelerator known as a cyclotron, which uses a constant magnetic field and constant-frequency electric field.
Proton therapy uses the protons from hydrogen atoms that are left behind after the electrons are removed. A cyclotron accelerates these protons then sends them through a beamtransport system to individual treatment rooms, where they are used to treat tumours. The proton beam can be contoured to the shape of the tumour, further decreasing radiation exposure to healthy cells and limiting side effects.




Red Bull makes hydrogen fuel cell play with AVL
Surely EVs are the best solution for motor sports and for weight / performance dispense with the battery altogether by introducing paired conductors...