The term phase refers to an arrangement of atoms in a substance which gives it a certain set of properties. The most familiar phases are the three states of matter: solid, liquid and gas. But particularly in solids, there can be many more phases, characterised by distinctive crystal structures that lead to quite different materials. Ice, for example, has 10 distinct phases; there are many different phases of steel, and piezoelectric materials gained their ability to generate flows of current by switching between different phases in response to external stimuli.
A team from the University of Pennsylvania has discovered previously unknown phases "hiding" in the compound strontium titanate (SrTiO3). Used in optical instruments, this material was already known to have unusual phase properties: it has a symmetrical, non-polar crystal structure that can be induced to switch into a different, polar (fractionally charged) phase by placing opposingly-charged ions along the long axis of its crystal.
The discovery, which the team describes in Science, could be the key to creating materials with properties that can be changed in trillionths of a second like flicking a switch or changing the state of a digital circuit.
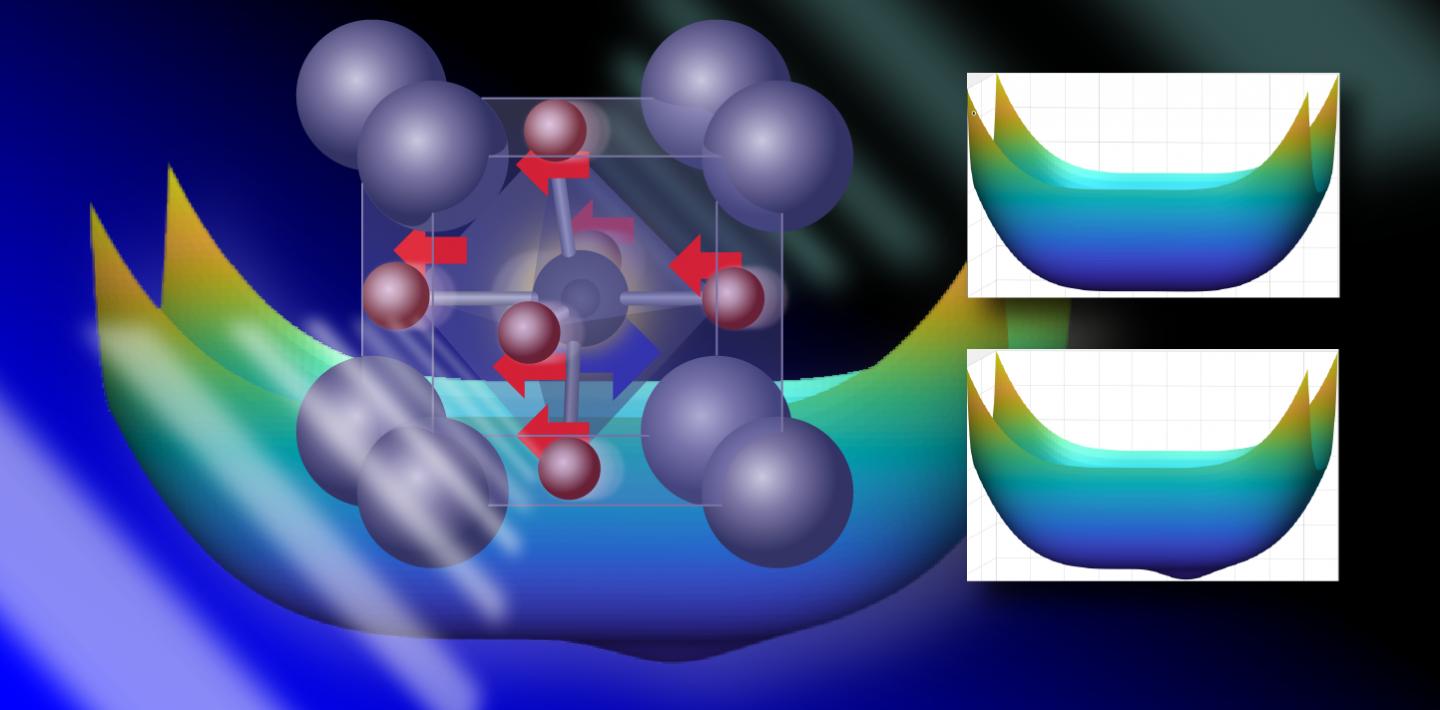
The team, a collaboration between physics theorists led by Andrew Rappe and experimentalists led by Keith Nelson, built on previous work using light to induce phase transitions in solid materials and how this could be used to develop atomic-level computer models. With Rappe creating a computer model of the structure of SrTiO3 with every atom and its movement represented, and Nelson carrying out experiments, the team discovered that when SrTiO3 is excited with light, the ions that make up its structure are pulled in different directions. But rather than falling back to the previous position when the light is switched off, vibrational movements in surrounding atoms lock the shifted ions into their new position, preserving the properties of this distinct structure, which separates positive and negative charges in space: a property which can be used to create capacitors with tunable capacitance, useful in electronics
The team compares this to pushing a pendulum, but rather than the pendulum swinging back, a small notch in the mechanism keeps it in place. Going back and forth between Rappe’s models and Nelson's experiments, the collaborating teams found experimental evidence that the theory Rappe had developed about this phase-switching behaviour was true.
"It's been a really awesome collaboration," says Nelson. "And it illustrates how ideas can simmer and then return in full force after more than 10 years."
The next phase of the collaboration will see the teams trying to replicate the phase-changing properties of SrTiO3 in new materials, and investigating whether light-pulse phase changes could be generated to be longer lasting, and whether properties such as insulators that change to metallic conductors or magnetic materials that switch between different magnetic polarities might be possible. This would create nanomaterials with useful applications in electronics and other fields, they claim.
"It's the dream of every scientist: To hatch an idea together with a friend, to map out the consequence of that idea, then to have a chance to translate it into something in the lab, it's extremely gratifying. It makes us think we're on the right track towards the future," says Rappe.




Swiss geoengineering start-up targets methane removal
No mention whatsoever about the effect of increased methane levels/iron chloride in the ocean on the pH and chemical properties of the ocean - are we...