The use of implanted structures and devices in medicine has become increasingly common in recent decades. However, the biggest problem in this field has been and remains the body's ability to recognise and reject foreign material.
While physicians want their implants to be accepted by the body and – in the case of joint replacements, for example – incorporated into the natural structures, poor integration is a constant risk. Moreover, if infection sets in around the implant, the increasing prevalence of antibiotic resistant bacteria means that the implant often has to be removed, necessitating more risky surgery.
A team from the Applied Plasma Physics and Surface Engineering Laboratory at the University of Sydney has now developed a method for attaching custom-made peptide molecules – essentially short fragments of proteins – to the surface of implants. This coating, they claim, provides biological signals to the surrounding tissue that suppress the immune response and encourage good integration.
In a paper in Nature Communications, the team explains that 10nm-long peptides are preferable to full-length proteins because they are more resistant to sterilisation, and therefore their full functionality survives the preparation necessary before an implant can be emplaced. Moreover, they can be made more easily by chemical synthesis.
“Although proteins have successfully been used in a number of applications, they don’t always survive harsh sterilisation treatments - and introduce the risk of pathogen transfer due to their production in micro-organisms,” said Prof Marcela Bilek, who led the research.
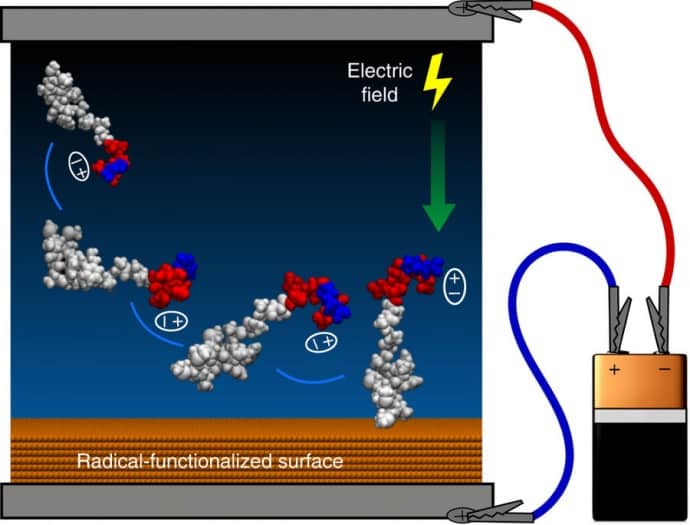
An important factor in the discovery is the way that the peptides are attached to the implant surface. This has to be through strong covalent bonds, because this prevents the molecules from being absorbed into surrounding tissues. To achieve this bond, Bilek’s team used an electric field to guide and attach the peptides to the surface. The density and orientation of the peptides on the surface are crucial to the devices acceptance by the body.
The surface first had to be prepared for the peptide coating. The team achieved this by attaching a pre-coating known as a radical-functionalised plasma polymer containing carbon, nitrogen and oxygen. This was deposited onto a titanium surface, and bombarded with negative ions during the deposition to create a high concentration of unpaired electrons within the structure of the coating, particularly on the oxygen atoms. It is these electrons that form the covalent bonds with the peptides.
The peptides were specifically designed so that the structures forced a separation of electric charge between one end of the molecule and the other. An electric field, generated by a simple household battery, is sufficient to rotate the peptide molecule in a solution applied to the coated implant so that it is in the correct orientation and guided onto the surface where it can attach.
“The application of our approach ranges from bone-implants to cardiovascular stents and artificial blood vessels,” said Behnam Akhavan, a team member from the university’s School of Aerospace, Mechanical and Mechatronic Engineering. “For the bone implantable devices, for example, such modern bio-compatible surfaces will directly benefit patients suffering from bone fracture, osteoporosis, and bone cancer."
If industry support and funding for clinical trials is forthcoming, Akhavan believes that peptide-coated implants could be available in around five years.




Project to investigate hybrid approach to titanium manufacturing
Sadly they will not be ordering any more presses from Wilkins & Mitchell http://www.historywebsite.co.uk/articles/Darlaston/WM.htm