The problem of looking at the inside of the body is a pressing one for doctors, and one particularly difficult issue is the gastrointestinal tract. Cancers in this system — the oesophagus, stomach, intestines and rectum — are major causes of death and difficult to investigate, because two of the most useful diagnostic methods for looking inside the tract and extracting samples: gastroscopy, where a camera on a flexible probe is inserted down the throat; and endoscopy, where a similar system goes in the other end — are so unpleasant and traumatic for the patient. But help might be at hand, thanks to the humble tadpole.

The idea of placing a camera in a swallowable capsule that can navigate its own path through the digestive tract and send pictures back to doctors isn’t a new one: indeed, so-called capsule endoscopy currently the best way to get pictures of the intestines between the stomach and colorectal system, the boundary zone between gastroscopy and endoscopy. But it isn’t without drawbacks: The speed of transport through the system, from muscular ripples along the intestines called peristalsis can vary widely from person to person; it can be difficult to determine precisely where lesions and growths are, because of mismatches between imaging speed and peristalsis, which can also lead to unclear images; and in the parts of the system where the capsule is free to tumble, it can sometimes be pointing in the wrong direction to provide a picture of a site of concern.
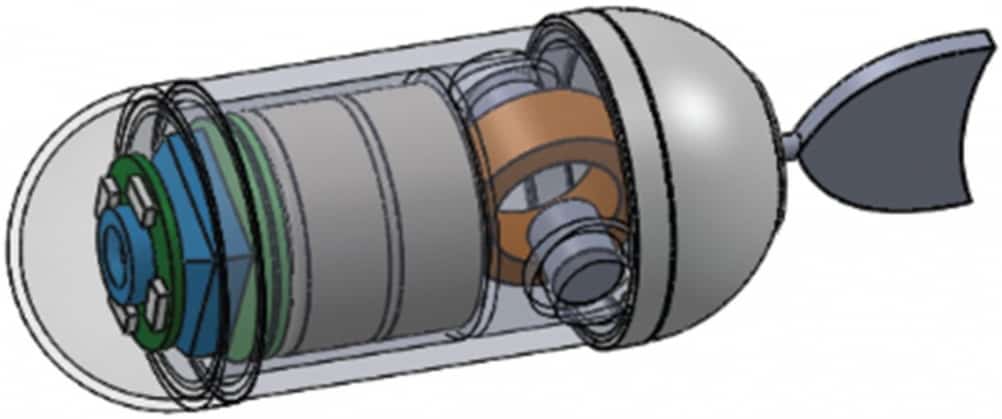
This is where the tadpole comes in, with researchers Yong Zhong and Ruxu Du from the Institute of Precision Engineering and Philip W Y Chiu of the Jockey Club Minimally Invasive Surgery Training Centre, both at the Chinese University of Hong Kong, attempting to mimic the movement of the larval amphibians to design a capsule endoscope with a controllable swimming action that doctors can steer around inside the stomach to provide a guided tour, then orient it ideally for its dive into the intestines where peristalsis can take over for the rest of its journey. So far they have produced the capsule, using 3D printing, and an experimental propulsion system, and tested it inside a pig’s stomach.
The capsule itself is a simple structure, 13mm in diameter, designed to hold a camera, LED light, an integrated circuit for receiving control instructions and transmitting data, antenna, 1.5V button battery and, at the rear, the drive unit, to which a rubber flapping tail is attached. This is much shorter compared with the body than a tadpole tail, to keep the overall capsule size small (its total length, with a 9mm tail, is 38mm; it weighs 6g) and reduce trauma to the patient. The drive unit consists of two permanent magnets situated on either side of (and perpendicular to) a magnetic coil, to which the tail is connected. The permanent magnets are placed so that their coils are in opposing directions, so that when the coil is energised, which genersates its own magnetic field, one of the permanent magnets attracts it while the other repels it. Oscillating the direction of current in the coil therefore causes the tail to flap back and forth. In liquid, this creates the thrust that propels the capsule along; varying the current changes the amplitude of the flapping and allows the capsule to be steered, as the researchers explain in this paper in the journal HKIE Transactions.
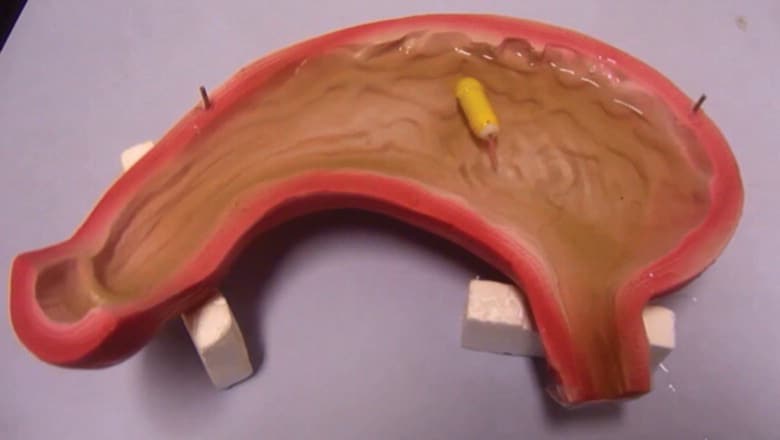
In tests using a mock-up double-scale stomach, the team determined the capsule’s maximum swimming speed is 12.5mm/sec and its minimum turning radius is 25mm. Further tests using a pig’s stomach proved that the capsule could be manoeuvred around using an IR remote control and turn when it reached the stomach wall. The researchers envisage that the patient would swallow the capsule on an empty stomach, then a clinician would steer it around to obtain images of the stomach. The patient would then be sent home with a sensor pad strapped to their abdomen while the capsule negiotiated the twists and turns of the intestine, returning to the hospital the following day for the clinician to retrieve images from the pad. “Future works include optimising the system model and supplementing more experiments to test it, integrating an image system into the capsule in order to complete the whole system, and finally in vivo testing in animals,” the team says.




Swiss geoengineering start-up targets methane removal
No mention whatsoever about the effect of increased methane levels/iron chloride in the ocean on the pH and chemical properties of the ocean - are we...