A low-cost ventilator developed by a team at the University of Glasgow, could help keep seriously ill people alive long enough to either recover or receive hospital care it is claimed.
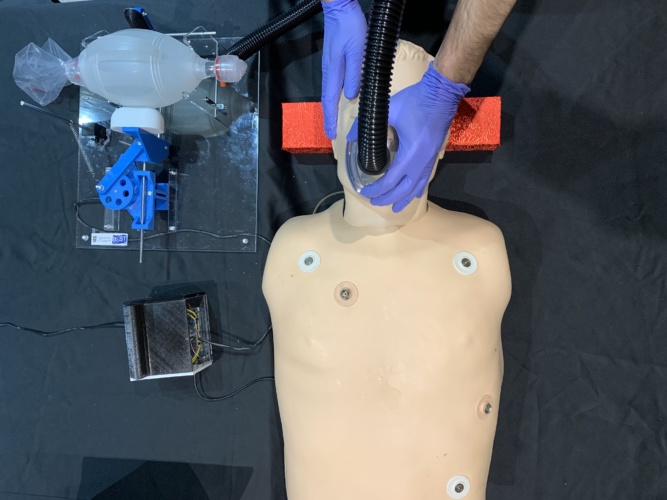
The device, called GlasVent, was developed by the University’s Professor Ravinder Dahiya and a team of engineers from his Bendable Electronics and Sensing Technologies (BEST) group.
The team began working on the project in mid-March, during the early stages of the UK’s COVID-19 outbreak, when demand for ventilators was expected to outstrip demand within weeks. Although these fears weren’t realised, the team claims that manufacture of its low cost system could be rapidly scaled to allow basic ventilatory support for large numbers of patients if coronavirus infections peak again, or if other pandemics break out in the future.
The system is effectively an automated version of the bag valve mask, a manually operated balloon like device which is already commonly used in emergency medical situations to deliver air into a patient’s lungs.
The team has developed a way to automate this simple device by using an Arduino microcontroller to control the motion of a 3D-printed slide-crank that provides a squeezing action on the sides of the bag to deliver oxygen to the patient.
The team has outlined plans for three GlasVent variants including a fully manual version, a mains-powered version, and a battery-powered version. According to the group, the build cost for these would range from £35 to £135, potentially making the system ideally for use in developing world countries.
More COVID-19 technology news from The Engineer
Commenting on the project Prof Dahiya said: “When the seriousness of the coronavirus pandemic started to become clear, my research group and I were keen to do whatever we could to help save lives. We’re proud that we’ve managed to go from design to build to testing in a matter of weeks. We’ve already conducted numerous successful tests on a medical mannequin fitted with artificial lungs, provided by the Royal Alexandra Hospital Paisley, so we’re confident that it is fit for purpose.
The team is currently working towards getting regulatory approval for the device to be used in clinical settings and are exploring options for commercial partnerships to manufacture the devices.




Red Bull makes hydrogen fuel cell play with AVL
Surely EVs are the best solution for motor sports and for weight / performance dispense with the battery altogether by introducing paired conductors...