Rechargeable lithium-ion batteries can lose energy over time and under certain conditions they overheat while working or charging, which can degrade battery life and reduce miles per charge.
The Engineer Q&A: Battery technology and the future of electrification
Powering up a British battery boom
Now, Nottingham University is collaborating with six scientific research institutes across China to develop an energy store with the combined performance merits of a solid-oxide fuel cell and a metal-air battery. It is claimed the new battery could significantly extend the range of electric vehicles, while being fully recyclable, environmentally friendly, low-cost, and safe.
Solid-oxide fuel cells convert hydrogen and oxygen into electricity because of a chemical reaction. They are highly efficient at extracting energy from a fuel, durable, low-cost, and greener to produce, but they are not rechargeable. Metal-air batteries are electrochemical cells that use a cheap metal such as iron and the oxygen present in air to generate electricity. During charging, they emit oxygen into the atmosphere. Although not very durable, these high-energy dense batteries are rechargeable and can store and discharge as much electricity as lithium-ion batteries, but much more safely and cheaply.
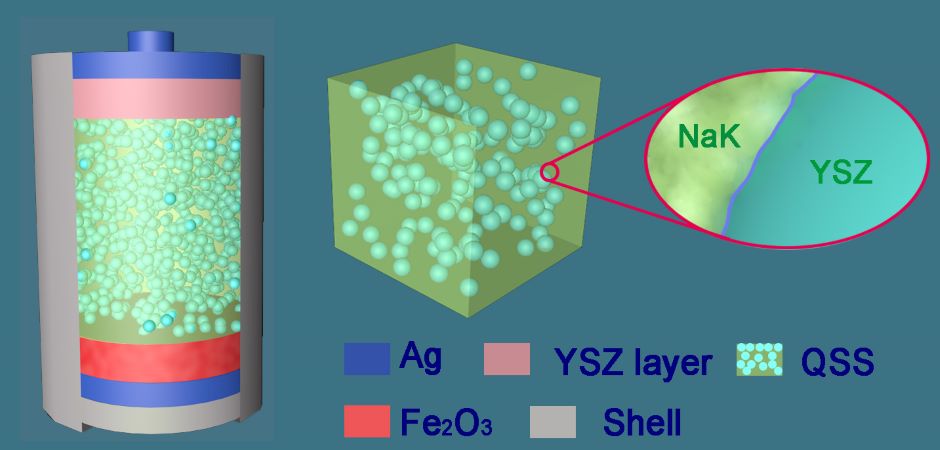
In the early research phases, the research team explored a high-temperature, iron-air battery design that used molten salt as a type of electrolyte - activated by heat - for electrical conductivity. Cheap and inflammable, molten salts help to give a battery impressive energy storage and power capability and a favourable lifecycle. However, molten salts also possess adverse characteristics.
In a statement, Professor George Chen, Nottingham University study lead, said: “In extreme heat, molten salt can be aggressively corrosive, volatile and evaporate or leak, which is challenging to the safety and stability of battery design. There was an urgent need to fine-tune these electrolyte characteristics for better battery performance and to enable its future use in electric transport.”
The researchers have now successfully improved the technology by turning the molten salt into soft-solid salt, using solid oxide nano-powders. Professor Jianqiang Wang, from the Shanghai Institute of Applied Physics, Chinese Academy of Sciences, who is leading this collaboration project has predicted that this quasi-solid-state (QSS) electrolyte is suitable for metal-air batteries which operate at 800ºC as it suppresses the evaporation and fluidity of the molten salts that can occur at such high operating temperatures.
Prof Wang’s colleague, Dr Cheng Peng said the quasi-solidification has been achieved using nanotechnology to construct a flexibly connected network of solid oxide particles. These act as a structural barrier, locking in the molten salt electrolytes while still allowing them to conduct electricity in extreme heat.
Professor Chen, who is leading a molten salt electrolysis laboratory in Nottingham, hopes the team’s “encouraging results” will help to establish a simpler and more efficient approach to designing low-cost and high-performance molten salt metal air batteries with high stability and safety.
“The modified molten salt iron-oxygen battery has great potential applications in new markets, including electric transport and renewable energy which require innovative storage solutions in our homes and at grid-level,” he said. “The battery is also, in principle, capable of storing solar heat as well as electricity, which is highly-desirable for both domestic and industrial energy needs. Molten salts are currently used at large scale in Spain and China to capture and store solar heat which is then converted to electricity - our molten salt metal air battery does the two jobs in one device.”
The team’s findings are published in a paper - ‘Quasi-solid-state electrolyte for rechargeable high-temperature molten salt iron-air battery’ - in Energy Storage Materials.

Hard hat mounted air curtain adds layer of protection
Something similar was used by miners decades ago!