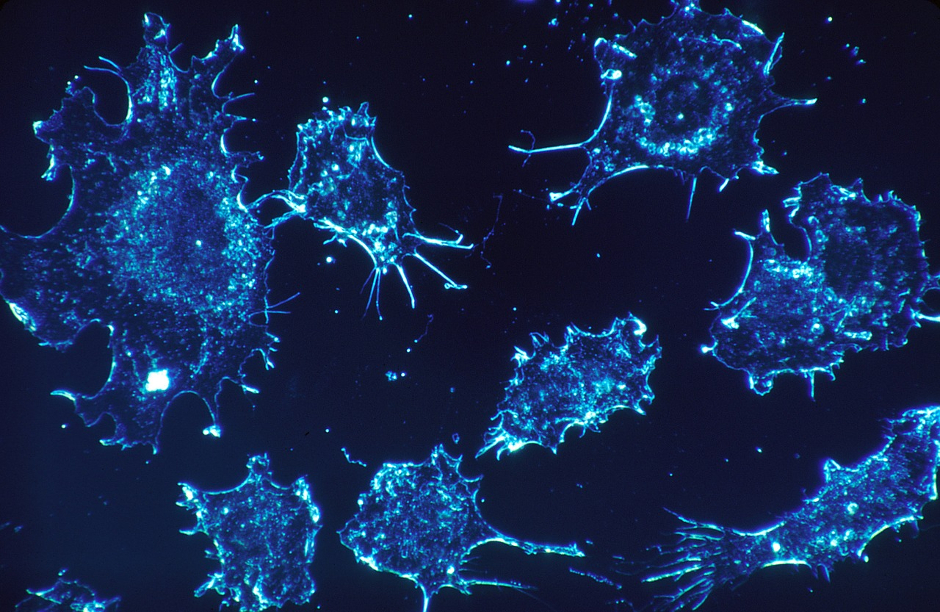
Project AMPLITUDE (Advanced Multimodal Photonics Laser Imaging Tool for Urothelial Diagnosis in Endoscopy) is developing a scanner to provide instant, non-invasive, precise and detailed information to determine what stage and grade that a tumour has reached. The project is coordinated by Tampere University, Finland and includes contributions from Aston University and Edinburgh-based Modus Research and Innovation.
C2I2019: Cancer probe promises more effective surgery
Cancers that form in the bladder are difficult to spot in their early stages and are often missed when using white light. At present, clinicians struggle to create accurate, detailed images inside the bladder because light cannot penetrate deep into the tissue.
Currently, tests for bladder cancer involve urinalysis (a check for blood in the urine), urine cytology (using a microscope to search for cancer cells in urine), or urine tests for tumour markers (a test to see if a patient has chromosome changes that are often seen in bladder cancer cells or higher levels of a protein called NMP22).
These tests can find cancerous cells in urine but they do not catch the disease in its early stages and sometimes the tests can miss biomarkers altogether.
Using multi-wavelength lasers, the new system will reportedly create an image from deep inside the tissue to give a quick and accurate diagnosis of the presence of a tumour, as well as its stage and grade.
The AMPLITUDE project combines medical and physics expertise to develop a new multi-modal imaging system complete with an endoscopic probe that delivers an instant diagnosis in a clinical setting.
In a statement, Project coordinator, Dr Regina Gumenyuk said: “A societal challenge for the next few decades is the instant diagnosis of major diseases.
“Photonics provides excellent opportunities to give healthcare professionals advanced, non-invasive diagnostics that detect symptoms and diseases at an early stage.
“The multimodal imaging approach in AMPLITUDE is based on three modalities, which together will provide precise and detailed information necessary to determine the tumour stage and grade.
“The system being developed by AMPLITUDE will be the first device to deliver a label-free procedure. This means we can avoid using fluorophores and their phototoxic effects which can sometimes damage cells.
“A reduction in the phototoxicity during autofluorescence imaging will minimise cell damage.”
The system uses infrared light to visualise deep inside the tissue, allowing scientists to peer into 'biological windows’ or infrared areas where light penetration increases in depth.
"The so-called 'third biological window’ - a range of 1550–1870nm - has not yet been extensively investigated,” said Dr Gumenyuk. “While it is possible to investigate this range at present, we would need a Supercontinuum laser source, which is prohibitively expensive. The new lasers that will be developed by AMPLITUDE will allow us to explore this wavelength range with compact and cost-effective systems.”

Hard hat mounted air curtain adds layer of protection
Something similar was used by miners decades ago!