Nanosensor detects pesticides on fruit in minutes
Pesticides can be detected on fruit in minutes thanks to new nanosensors developed by researchers at Karolinska Institutet in Sweden.
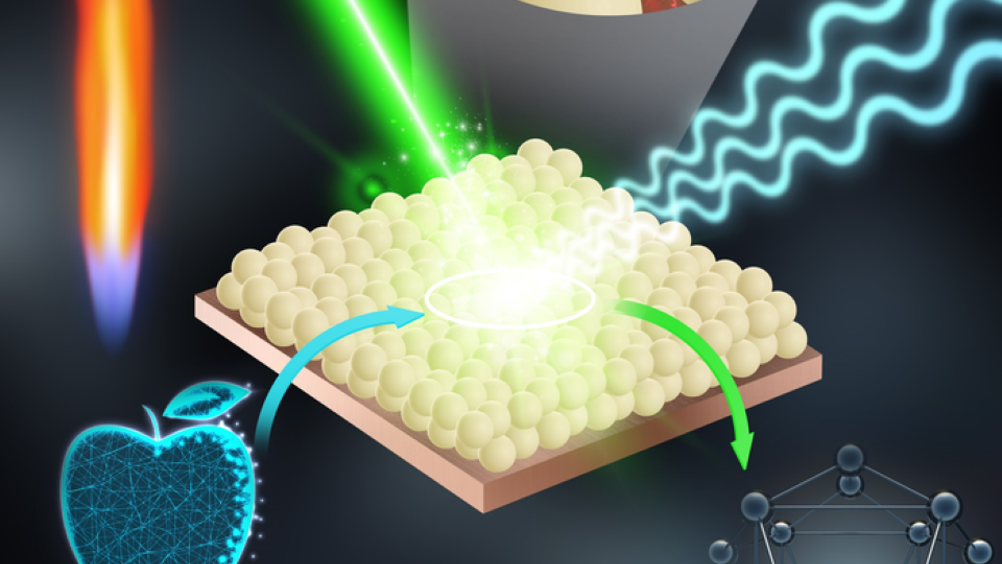
The proof-of-concept technique uses flame-sprayed silver nanoparticles to increase the signal of chemicals. While still at an early stage, the researchers hope these nano-sensors could help uncover food pesticides before consumption. The team’s findings are published in Advanced Science.
“Reports show that up to half of all fruits sold in the EU contain pesticide residues that in larger quantities have been linked to human health problems,” said Georgios Sotiriou, principal researcher at the Department of Microbiology, Tumour and Cell Biology, Karolinska Institutet, and the study’s corresponding author. “However, current techniques for detecting pesticides on single products before consumption are restricted in practice by the high cost and cumbersome manufacturing of its sensors. To overcome this, we developed inexpensive and reproducible nano-sensors that could be used to monitor traces of fruit pesticides at, for example, the store.”
The new nanosensors employ surface-enhanced Raman scattering (SERS), a sensing technique that can increase the diagnostic signals of biomolecules on metal surfaces by more than one million times. The technology has been used in several research fields, but high production costs and limited batch-to-batch reproducibility have hindered widespread application in food safety.
Register now to continue reading
Thanks for visiting The Engineer. You’ve now reached your monthly limit of news stories. Register for free to unlock unlimited access to all of our news coverage, as well as premium content including opinion, in-depth features and special reports.
Benefits of registering
-
In-depth insights and coverage of key emerging trends
-
Unrestricted access to special reports throughout the year
-
Daily technology news delivered straight to your inbox










UK Enters ‘Golden Age of Nuclear’
The delay (nearly 8 years) in getting approval for the Rolls-Royce SMR is most worrying. Signifies a torpid and expensive system that is quite onerous...