Researchers in Beijing, working on generating electricity from natural, involuntary movements of the human body, such as the contractions of their hearts, respiration and blood flow have developed a cardiac pacemaker which operates via a piezoelectric film covering the surface of the heart. Tested in a pig, whose heart is a similar size and has similar demands placed on it as a human’s, the device corrected faulty heart rhythm and prevented cardiac deterioration, the team claims. Such devices, which would not be limited to cardiac pacemakers, could represent a paradigm shift in medical implants.
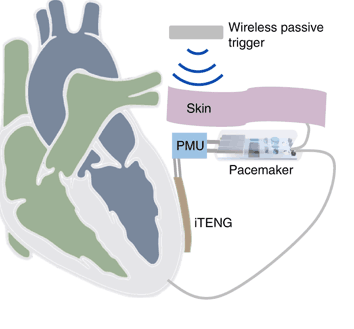
Currently, implanted electrical devices depend on batteries which tend to be bulky, rigid, limited in lifespan and, despite advances in reducing the energy demands of devices, still represent an impediment on the progress of implants. Attention has turned in recent years to using processes that occur in the body itself as a source of power. Involuntary movement is the most widely investigated of these, as it can be turned into electricity through the use of piezoelectric materials which generate a charge when deformed. One drawback that has been encountered is that such devices are often developed using animal models, and laboratory animals tend to be much smaller than humans and therefore require much less energy from the devices to power such functions as cardiac pacing.
The Chinese team, based at the Beijing Advanced Innovation Centre for Biomedical Engineering, stated in a paper in Nature Communications that it was inspired by biological symbiosis, a phenomenon whereby different organisms that live in close proximity to each other interact to their mutual benefit, such as bacteria which live in nodules on the roots of leguminous plants (clover, peas and beans) and allow them to make use of nitrogen in the soil, to develop their self-powering mechanism.
In this case, the mechanism behind the power generation is not in fact piezoelectricity but the tribological effect, where two materials rubbing against each other generate a charge. Most people will be familiar with this from the buildup of static urgency caused by walking on a synthetic fibre carpet or rubbing a balloon against hair and sticking it on a wall. In their paper, corresponding authors Zhong Lin Wang, Hao Zhang, Zhou Li and their colleagues describe a triboelectric energy generator consisting of three layers: a titanium ribbon as the "keel" or static layer, overlaid by a film of nanostructured PTFE and with a layer of an elastic sponge formed of EVA copolymer between the two. Additional materials helped to stabilise the assembly.
This generator was attached to a power management unit containing a chargeable capacitor and a standard commercial pacemaker attached to this. They implanted the device into a 45kg male pig, with the triboenergy generator placed between the left ventricle of the heart and the pericardium, the membrane that encloses the heart. Every time the heartbeat, the keel and PTFE layer of the generator were forced into contact and separated, generating a charge which was stored in the capacitor. When fully charged, the capacitor powered the pacemaker to produce pulses of 4V spaced 0.9ms apart.
The experiment was designed only to demonstrate the feasibility of such a device, and the charging period of around three hours and 20min for only one minute of pacemaker operation does not represent a realistic vision of how such a device might work in humans.
Commenting on the paper, Charles Pearman, a clinical lecturer in cardiovascular science at Manchester University stated: "The technology as it stands would therefore need to be significantly more efficient at extracting energy to be useful for those who require their pacemakers to function continuously.”
In another comment, Prof Tim Chico, an honorary consultant cardiologist at Sheffield University, noted that the device would require open-heart surgery to implant, which is far more invasive than the current techniques used for pacemaker installation. “However, the device could use movement from other muscles rather than the heart, so this should not be a problem,” he added. The major issue with the research is that it needs to demonstrate that the device can store enough electricity to power the pacemaker to operate most of the time, he added.




About:Energy tasked with McMurtry battery challenge
Zero reason to own an EV that has blistering performance if it can't travel more than a few miles before needing a recharge. I saw the McMurty EV run...