The technology is being developed at Purdue University, Indiana with the help of a $100,000 Grand Challenges Explorations grant from the Bill & Melinda Gates Foundation.
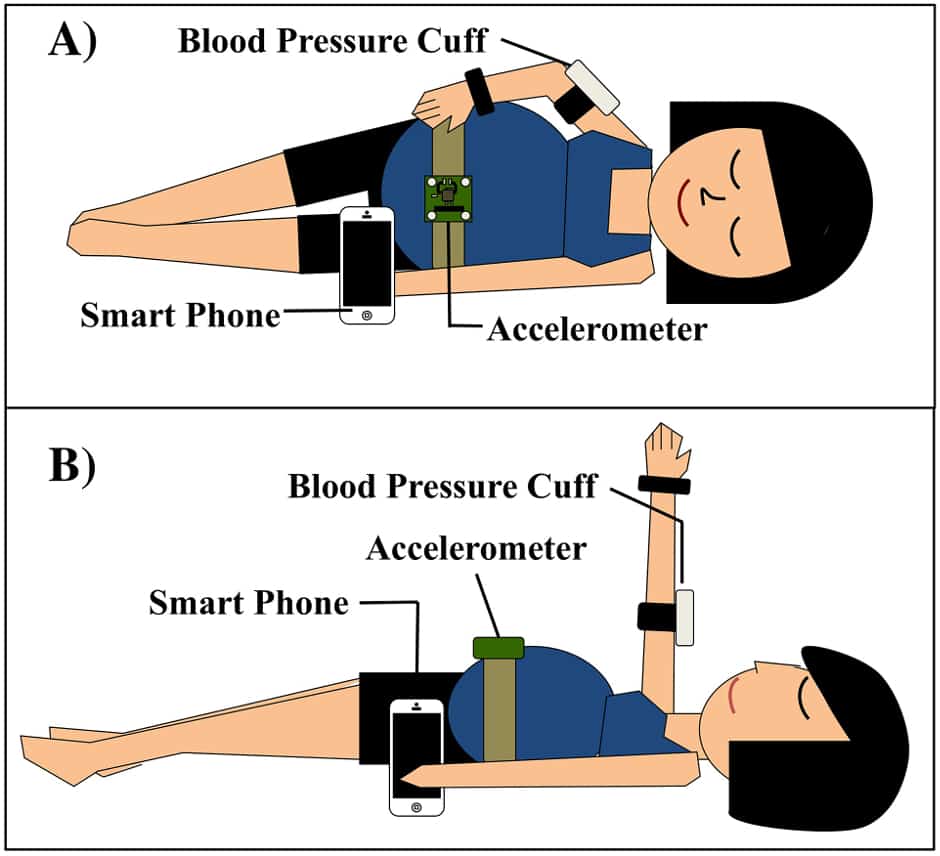
Led by Craig Goergen, an assistant professor in Purdue University’s Weldon School of Biomedical Engineering, the team is working to combine smartphones, a conventional inflatable blood pressure cuff, and a wireless accelerometer to build a prototype that detects preeclampsia before it develops.
According to Purdue, the most innovative aspect of the device is its use of the supine pressor test, which measures whether a woman’s blood pressure increases when she changes position from lying on her left side to lying on her back. If the diastolic pressure increases enough, it is a warning sign that a woman is susceptible to preeclampsia.
“This is a device that women are going to be able to use at home with a minimal amount of training,” Goergen said.
Women will send the results to a doctor's office, a health-care system or a centralised network for the results to be read and where they could receive counselling so they can start management and treatment options as early as possible.
While the Gates Foundation’s goal is to help women in developing countries, Goergen said the device could help women in inner cities and rural areas of the United States and other developed countries. They plan on initially testing the device on low- and middle-income women in and around Indianapolis once the researchers receive the institutional approvals.
“It will be a great way to make sure that these patients are not going down a road that is going to lead to problems for both them and their baby,” Goergen said.
The American Journal of Obstetrics & Gynecology published a report in 2017 estimating the costs to the US health-care system for preeclampsia at $2.18bn for the first 12 months after birth, split between $1.03bn for mothers, and $1.15bn for babies.
The research team – which includes Dr David Reuter of Seattle Children’s Hospital, George Wodicka, the Dane A Miller Head of Biomedical Engineering at Purdue, and Kirk Forster, a senior research engineer at the Weldon School - have obtained a provisional patent for their device. They are now looking for partners to help further develop the technology.





Plasma technique converts landfill methane to jet fuel
Trevor; there are still many landfill sites in the UK where the methane is not captured but not only that, where there is protein waste in them, they...