The development of a device meant to assist or completely replace functioning of the heart is undeniably complex. This design process involves immense challenges, from supplying power to the device to ensuring it does not interfere with normal biological functioning. Researchers at Abbott Laboratories use multiphysics simulation to engineer left ventricular assist devices (LVADs) in an ongoing effort to improve the outlook and quality of life for patients with heart failure.
The condition typically begins with the left side of the heart, as the left ventricle is responsible for pumping oxygen-rich blood throughout the body a greater distance than the right ventricle, which pumps blood through the lungs. Often, in patients with a poorly functioning left ventricle, an LVAD (see Figure 1) can provide mechanical circulatory support.

Figure 1. An LVAD pump is responsible for circulating oxygen-rich blood throughout the body. Image courtesy of Abbott Laboratories.
The ventricle assist device is one of the most complex machines ever implanted in a human being. An LVAD must circulate the entire human blood stream and support life, as well as be compatible with the internal environment of the human body. Thoratec, now part of Abbott, brought LVADs to a wide market in 2010, after years of clinical trials.
Designing a Powerful, Efficient and Hemocompatible Pump
The design of an LVAD must take many factors into consideration. The device must be small enough to connect to the heart and be made of compatible materials and geometry that permit the device to reside in the body without being rejected. Fluid dynamics, power supply and thermal management must also be considered. As multiple interacting physical effects must be accounted for at each area of development, multiphysics simulation is vital to the design process.
Freddy Hansen, Staff Research Physicist at Abbott, uses his expertise in physics and mathematical modelling to characterise complex implantable medical devices like LVADs before experimental studies.
Hansen has been using the COMSOL Multiphysics® software since 2011 and has since created more than 230 models that address a wide range of design challenges pertaining to the unique physics of artificial pumping devices.
“I use COMSOL Multiphysics every day, from proof-of-concept models to quite sophisticated simulations featuring detailed CAD geometries and coupled physics. I work with some complex models for months before I have taken all of the information I want from them.”
With each generation of LVADs introduced to market, improvements are made that contribute to enhanced safety and quality of life for the patient. Research and development efforts at Abbott are centred on improving biocompatibility, hemocompatibility and immunocompatibility, such that the device does not illicit an adverse immune response or interfere with other bodily systems.
Geometry and size of the device play an important role in its overall effectiveness. To implant the LVAD, the surgeon connects one end of the LVAD to the left ventricle and the other end to the ascending aorta (see Figure 2). If the device is smaller, it is less cumbersome and less likely to interfere with neighbouring organs or tissue. Simulation allows for the evaluation of changes in size or geometry of the LVAD design before implementation in a physical prototype.
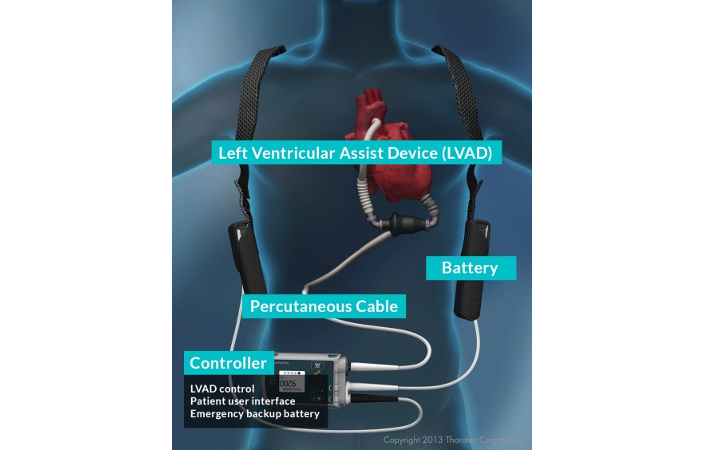
Figure 2. External equipment of an LVAD. Image courtesy of Abbott Laboratories.
Optimising LVAD Design for Biocompatibility
Many simulations were used in the development of the centrifugal pump of the LVAD. One challenge associated with engineering these devices is the prevention of blood clotting in any space in or around the pump. To address this, a magnetically levitated rotor was developed, which eliminated the need for ball bearings and other components with geometries that might promote clotting. Hansen used the Rotating Machinery modelling technology available in the software to model both the magnetically levitated rotor and turbulent fluid flow.
A permanent magnet in the pump rotor is driven by coils in the stator, which exert a torque on the rotor and provide active control of the position of the rotor axis. The vertical position — or levitation — of the rotor is accomplished by magnetic field line tension and does not need active control. The rotor receives blood axially and redirects it radially, into the volute, or fluid collector (see Figure 3). Some of the blood flows back and around the outer edge of the rotor and flows back into the rotor inlet, resulting in a constant washing of the blood, which serves to eliminate places where the blood can stagnate and clot.
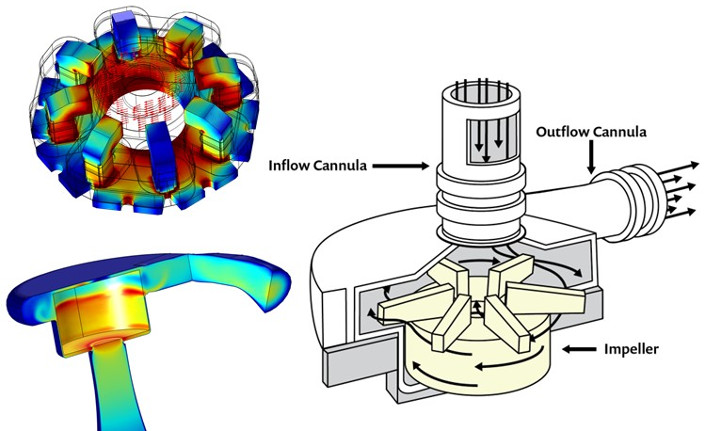
Figure 3. Visualisation of the magnetically levitated rotor, which eliminates the need for ball bearings and other components with geometries that might promote clotting. Here, the magnitude and direction of the magnetic field in the rotor, as well as the magnitude of the magnetic field in the stator are shown (top left). 3D CFD simulation depicting fluid velocity within the pump chamber (bottom left). Diagram of the centrifugal pump of an LVAD (right).
Another significant advance was the development of a pump system with pulsatile flow, rather than continuous flow, which more closely mimics a functioning heart. The pulsatile flow aids in the washing of the blood, preventing blood clots and is also believed to have a positive physiological effect on blood vessels throughout the body.
Wirelessly Powering a Fully Implantable LVAD
Current LVADs require power transfer from external batteries in a controller outside the body to the pump by way of a cable, made with materials engineered to be biocompatible. But what if the cable could be eliminated?
Hansen explored transferring power by way of magnetic resonance coupling. Magnetic resonance coupling occurs when two objects with almost the same resonance frequency transfer energy to each other through their oscillating magnetic fields. In this way, power can be transferred from a power source to another device, even through a biological medium such as tissue.
A fully implantable LVAD system (FILVAS) would decrease infection risk and improve patient quality of life, as the patient would not need to be concerned with cable management. With this concept, the patient could shower or swim without concern for the cable.
To assess the feasibility of wireless power transfer to an LVAD and determine how much power could be delivered between reasonably sized coils, Hansen coupled a 3D magnetic field model with an electrical circuit model to determine operating efficiency and power loss, as well as optimal circuit design and component values.
He evaluated different materials for important components, such as the wires of the transformer coils. He also studied the misalignment of a coil due to patient walking, running and other activities, and together with the effect of the presence of nearby magnetic or metallic objects.
Engineers also had to ensure that body temperature and biological systems will not be affected by the implant. The wireless transfer of energy induces small currents in the body tissue near the coils. Hansen modelled the heat generated in the tissue as a result of the induced currents; combined this with models of heat generated inside the implant (in magnetic wires, electronics and batteries) and then used the thermal conductivity coefficient determined from a famous Cleveland Clinic experiment, to determine the temperature increase in body tissue near the implant (see Figure 4).
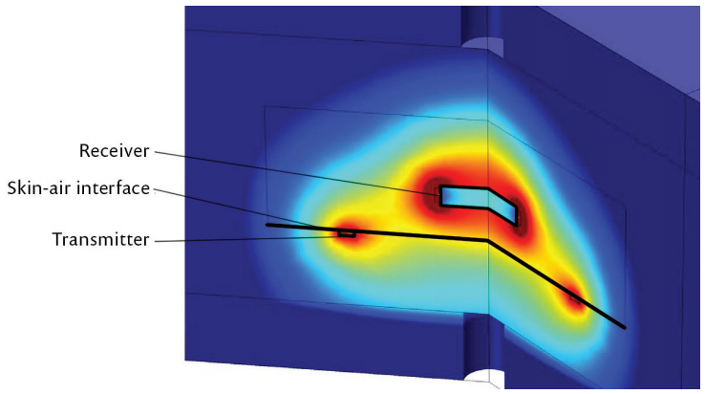
Figure 4. Model of heating induced in the body through magnetic power transfer. Results show power density distribution in the tissue and the surrounding air.
Protecting Life-Sustaining Batteries
Patients must live with their LVADs every single day, which inevitably means that the external LVAD controller must be able to withstand the wear and tear of life, as well as the occasional dropping of the controller to the floor. To ensure that the controller (which contains crucial life-saving batteries) will continue to function even if the patient tosses it around, Hansen developed a mechanical impact analysis of the controller in order to assess its resilience (see Figure 5).
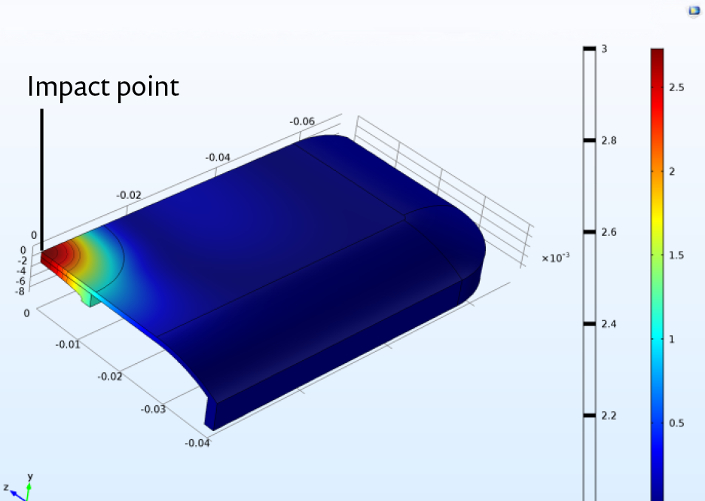
Figure 5. Simulation of a steel ball impacting an LVAD controller to evaluate resiliency of the controller. Visualisation of displacement along the vertical axis.
He also analysed the edges and surfaces of the deformed structural shell and the frame for twisting, to verify the integrity of the controller. The analysis proved that the controller would continue to provide life-sustaining power to the LVAD even after a substantial impact.
New Technology Shows Improved Options for Patients in the Future
In designing devices to assist and replace the function of the heart, multiphysics analysis has proven to be essential. Hansen combines experimental characterisation and mathematical modelling to understand the physics pertaining to ventricle assist devices and improve the biocompatibility of the device as well as the overall patient experience.
The latest innovations to mechanical pumping systems — including a smaller device size, a more hemocompatible pump, the introduction of pulsatile flow and now the possibility of wireless power transfer — hold much promise for better treatment in the future.

Freddy Hansen, PhD, Staff Research Physicist, Abbott Laboratories




Poll: Should the UK’s railways be renationalised?
All public service companies should be nationalised for many different reasons, particularly railways, not the least because the tax payer has already...