MIP sensor seeks antimicrobial resistant bacterial strains
Researchers at Newcastle University are investigating the development of a technique to detect antimicrobial resistant bacterial strains using special polymers designed to bond to bacteria.

The technique involves using molecularly imprinted polymers (MIPs), also known as polymeric antibody-mimics because of their ability to bond to a target molecule.
They are made from monomers which resemble amino acid structures and interact with bacteria in similar ways to antibodies, said Dr Marloes Peeters of the Advanced Materials group at Newcastle University School of Engineering, and principal investigator for the EPSRC-funded project. The polymers can be tailored to give them an affinity to bond to a particular target, such as a strain of bacteria that has developed resistance to antibiotics.
Micro-needle skin patches could help fight antibiotic resistance
Rapid-readout bacteria test can detect antibiotic resistance
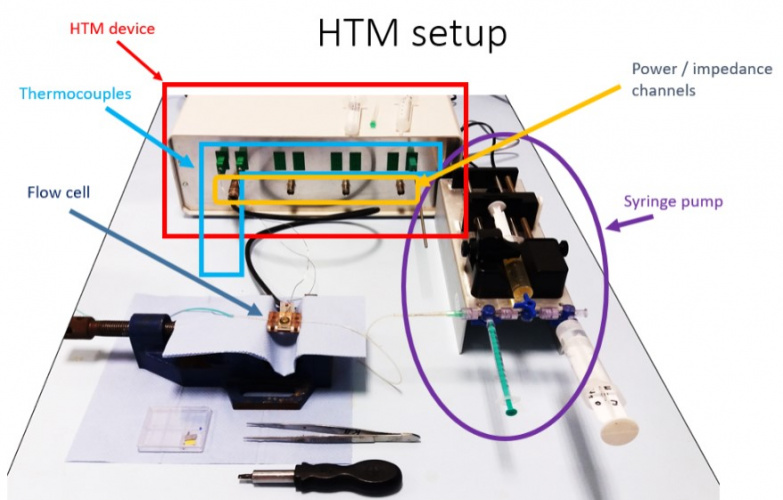
In previous work Dr Peeters’ group had demonstrated that the binding of the target to an MIP altered the conduction of heat through the polymer, leading to a temperature differential that is dependent on the concentration of the target bacteria. This can be measured in the lab by inserting the sensor into a flow cell, connected to a thermal sensor (a thermocouple device). This has been patented as the Heat Transfer Method.
Register now to continue reading
Thanks for visiting The Engineer. You’ve now reached your monthly limit of news stories. Register for free to unlock unlimited access to all of our news coverage, as well as premium content including opinion, in-depth features and special reports.
Benefits of registering
-
In-depth insights and coverage of key emerging trends
-
Unrestricted access to special reports throughout the year
-
Daily technology news delivered straight to your inbox










Water Sector Talent Exodus Could Cripple The Sector
Maybe if things are essential for the running of a country and we want to pay a fair price we should be running these utilities on a not for profit...