Spinal stimulators could give sensory feedback to prosthetic arms
Spinal stimulators used to relieve chronic pain could provide sensory feedback to a prosthetic arm, claim researchers at the University of Pittsburgh's Rehab Neural Engineering Labs.
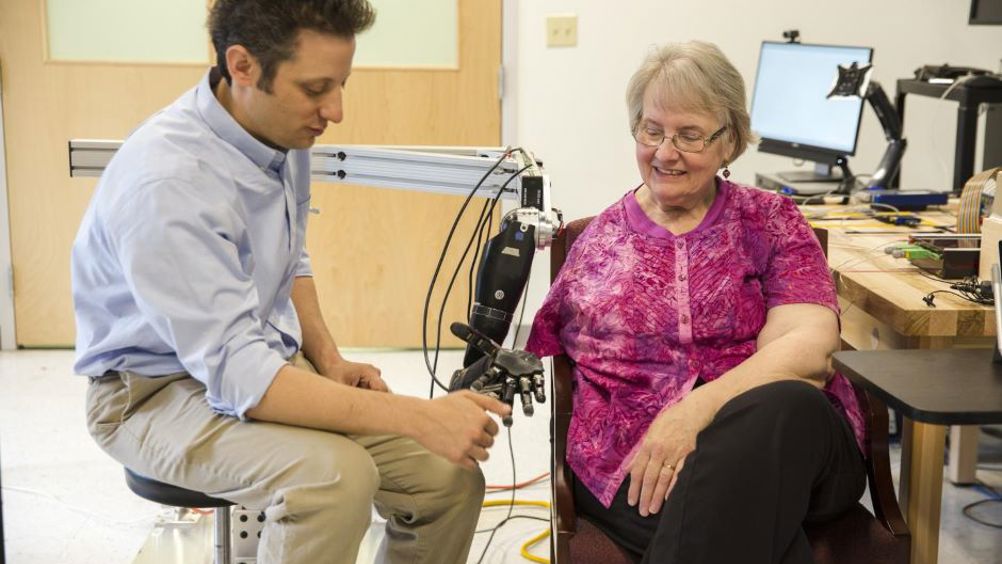
For their study, published in eLife, four amputees received spinal stimulators which create the illusion of sensations in the missing arm.
Future prosthetic: towards the bionic human
Next-generation bionic hand restores sense of touch
"What's unique about this work is that we're using devices that are already implanted in 50,000 people a year for pain - physicians in every major medical centre across the country know how to do these surgical procedures - and we get similar results to highly specialised devices and procedures," said study senior author Lee Fisher, Ph.D., assistant professor of physical medicine and rehabilitation, University of Pittsburgh School of Medicine (UPMC).
The strings of implanted spinal electrodes run along the spinal cord, where they sit slightly to one side, upon the same nerve roots that would normally transmit sensations from the arm.
Fisher's team sent electrical pulses through different spots in the implanted electrodes while participants used a tablet to report what they were feeling and where.
Register now to continue reading
Thanks for visiting The Engineer. You’ve now reached your monthly limit of news stories. Register for free to unlock unlimited access to all of our news coverage, as well as premium content including opinion, in-depth features and special reports.
Benefits of registering
-
In-depth insights and coverage of key emerging trends
-
Unrestricted access to special reports throughout the year
-
Daily technology news delivered straight to your inbox










Water Sector Talent Exodus Could Cripple The Sector
Maybe if things are essential for the running of a country and we want to pay a fair price we should be running these utilities on a not for profit...