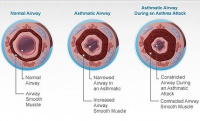
In use, the company’s Alair Bronchial Thermoplasty System delivers thermal energy to the airway wall in a precisely controlled manner to reduce excessive airway smooth muscle.
Airway smooth muscle is located within the walls of airways in the lung and the excessive and inappropriate constriction of the muscle is a predominant feature of asthma. Reducing airway smooth muscle decreases the ability of the airways to constrict, thereby reducing the frequency of asthma attacks.
The bronchial thermoplasty treatment is performed using a flexible bronchoscope that is introduced through a patient’s nose or mouth, and into their lungs. The tip of a small-diameter catheter is then expanded to contact the walls of targeted airways. Controlled thermal energy is then delivered to the airway walls to reduce the presence of the excess airway smooth muscle.
The minimally invasive procedure, like many other flexible endoscopy procedures, is done under moderate sedation and the patient typically returns home the same day.
By decreasing the ability of the airways to constrict, this new treatment has been shown to help patients with severe asthma gain substantially better control over their disease.
FDA approval of the Alair System was largely based on the promising results of trials that demonstrated that patients treated with the Alair System have an improved quality of life over patients who relied solely on medical therapy; the patients also experienced other clinically significant benefits, including a 32 per cent reduction in asthma attacks.





IEA report claims batteries are ‘changing the game’
It is important in trucks and other commercial vehicles as the payload will be reduced if the battery weight is penal. Battery applications in the...