US engineers "print" functioning artificial ear
Scientists at Princeton University in the US used off-the-shelf printing tools to create a functional ear that can ‘hear’ radio frequencies beyond the range of normal human capability.
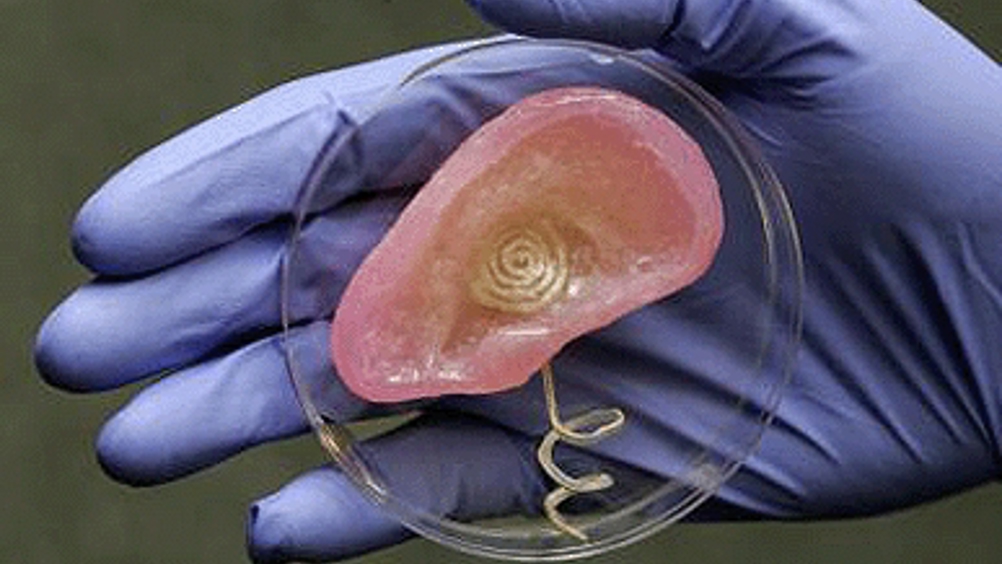
The researchers’ primary purpose was to explore an efficient and versatile means to merge electronics with tissue. The scientists used 3D printing of cells and nanoparticles followed by cell culture to combine a small coil antenna with cartilage, creating what they term a bionic ear.
‘In general, there are mechanical and thermal challenges with interfacing electronic materials with biological materials,’ said Michael McAlpine, an assistant professor of mechanical and aerospace engineering at Princeton and the lead researcher. ‘Previously, researchers have suggested some strategies to tailor the electronics so that this merger is less awkward. That typically happens between a 2D sheet of electronics and a surface of the tissue. However, our work suggests a new approach - to build and grow the biology up with the electronics synergistically and in a 3D interwoven format.’
Last year, a research effort led by McAlpine and Naveen Verma, an assistant professor of electrical engineering, and Fio Omenetto of Tufts University, resulted in the development of a ‘tattoo’ made up of a biological sensor and antenna that can be fixed to the surface of a tooth.
Register now to continue reading
Thanks for visiting The Engineer. You’ve now reached your monthly limit of news stories. Register for free to unlock unlimited access to all of our news coverage, as well as premium content including opinion, in-depth features and special reports.
Benefits of registering
-
In-depth insights and coverage of key emerging trends
-
Unrestricted access to special reports throughout the year
-
Daily technology news delivered straight to your inbox










Water Sector Talent Exodus Could Cripple The Sector
Maybe if things are essential for the running of a country and we want to pay a fair price we should be running these utilities on a not for profit...